The iodine–starch test is a chemical reaction that is used to test for the presence of starch or for iodine. The combination of starch and iodine is intensely blue-black.
Purpose:
To detect the presence of starch in a given substance using the iodine test.
Principle:
Iodine reacts with starch to form a blue-black color complex. This reaction is used to identify the presence of starch in a given substance.
Requirements:
- Iodine solution (0.1% w/v) – 1 mL
- Potassium iodide solution (2% w/v) – 2 mL
- Distilled water – 97 mL
- Starch solution (1% w/v) – 1 mL
- Test Tube
- Dropper
Procedure:
- Prepare the iodine reagent by adding 1 mL of iodine solution and 2 mL of potassium iodide solution to 97 mL of distilled water in a clean, dry container.
- Mix the reagent well by stirring or swirling the container.
- Prepare the test sample by adding 1 mL of the starch solution to a clean, dry test tube.
- Add 2-3 drops of the iodine reagent to the test tube containing the starch solution.
- Observe the color of the solution. If the solution turns blue-black, then starch is present. If the solution remains yellow-brown, then starch is not present.
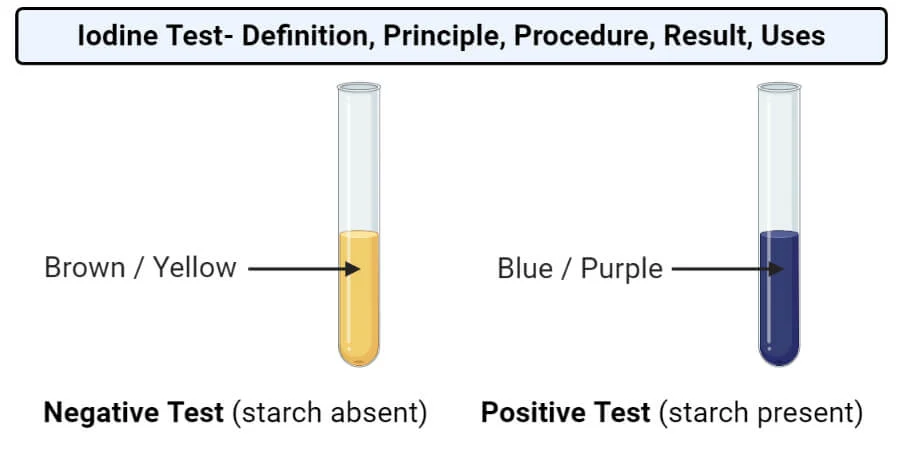
Results and Interpretation:
- If the test sample contains starch, the mixture will turn blue-black in color. This indicates a positive test for the presence of starch.
- If the sample does not contain starch, there will be no color change, indicating a negative test.
The iodine test is a simple and reliable method for detecting the presence of starch in a given sample
