The lipids solubility test is done to show the presence of lipids in a substance. The substance is first dissolved in ethanol. This solution is then dissolved in water. If lipids are present in the mixture, it will precipitates and forms an emulsion.
Purpose:
To determine the solubility of lipids in different solvents.
Principle:
Lipids are hydrophobic molecules that are insoluble in water but soluble in organic solvents such as ethanol, chloroform, and ether. The solubility of lipids can be used to distinguish them from other biomolecules such as proteins and carbohydrates.
Requirements:
- Samples of lipids (e.g., vegetable oil, butter, egg yolk, etc.)
- Test tubes
- Ethanol
- Chloroform
- Ether
- Water
Procedure:
- Take 01 ml of each sample (e.g., vegetable oil, butter, egg yolk, etc.) in 4 different test tubes.
- Add 1 ml of ethanol to the first test tube and shake it gently.
- Observe if the lipid dissolves or not. Record the result as “soluble” or “insoluble.”
- Repeat the same procedure with chloroform and ether.
- Add 1 ml of water to the last test tube and shake it gently.
- Observe if the lipid dissolves or not. Record the result as “soluble” or “insoluble.”
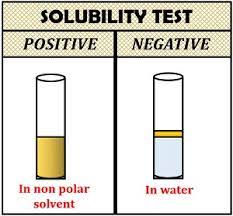
Results and Interpretation:
Lipids are generally soluble in organic solvents such as ethanol, chloroform, and ether but insoluble in water. The results of the test are interpreted as follows:
Sample | Ethanol | Chloroform | Ether | Water
Vegetable oil | Soluble | Soluble | Soluble | Insoluble
Butter | Soluble | Soluble | Soluble | Insoluble
Egg yolk | Soluble | Soluble | Soluble | Insoluble
The above table shows that all the lipid samples are soluble in organic solvents but insoluble in water. Therefore, the solubility test is a useful method to differentiate lipids from other biomolecules
Leave a Reply